Heberon® Alfa R
- Drug containing recombinant human interferon alpha 2b
- Heberon® Alpha R is an important modifier of the biological response.
- It is antiviral, antiproliferative and immunomodulatory.
- It offers an excellent therapeutic response to viral and immunological diseases and neoplasms.
- Its antifibrotic and antiangiogenic effects have been demonstrated and it acts against diseases of the central nervous system.
- The albumin-free forms of Heberon®Alfa R do not contain blood products and are less immunogenic.
Lyophilized powder for intramuscular, intravenous, subcutaneous, intralesional, intraperitoneal and intrathecal injection. Solution for intramuscular, intravenous, subcutaneous, intraperitoneal and intrathecal injection.
3 000 000 IU; 5,000,000 IU; 10,000,000 IU
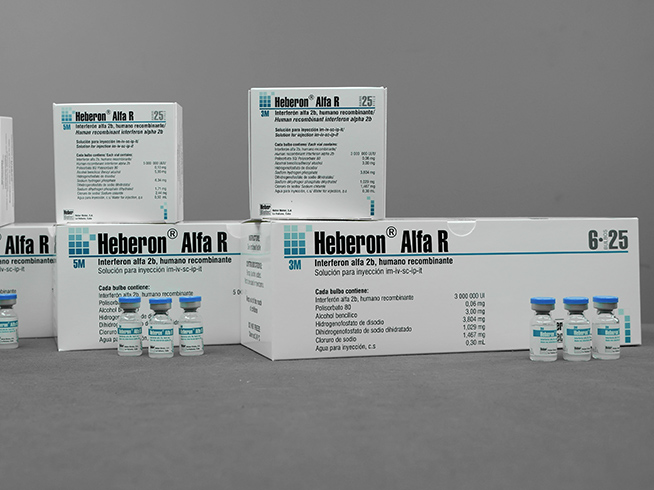
- Heberon® Alfa R 3 M
- Case for 1 or 25 colorless glass bulbs with 0.3 mL
- Case with 10 individual cases for 1 glass bulb colorless with 0.3 mL
- Case with 6 cases for 25 colorless glass bulbs with 0.3 mL
- Heberon® Alfa R 5 M
- Case for 1 or 25 colorless glass bulbs with 0.5 mL
- Case with 10 individual cases for 1 colorless glass bulb with 0.5 mL
- Case with 6 cases for 25 bulbs of colorless glass with 0.5 mL
- Heberon® Alfa R 10 M
- Case for 1 or 25 colorless glass bulbs with 1 mL
- Case with 10 cases individual per 1 colorless glass bulb with 1 mL
- Case with 6 cases for 25 colorless glass bulbs with 1 mL
Heberon® Alfa R 3 M (lyophilized powder): each bulb contains 3,000,000 IU recombinant human interferon alfa 2b; disodium hydrogen phosphate; sodium chloride; human serum albumin; dextran 40; mannitol 20%; Sodium dihydrogen phosphate dihydrate.
Heberon® Alfa R 5 M (lyophilized powder): each bulb contains 5,000,000 IU recombinant human interferon alfa 2b; disodium hydrogen phosphate; sodium chloride; human albumin 20%; dextran 40; mannitol 20%; Sodium dihydrogen phosphate dihydrate.
Heberon® Alfa R 10 M (lyophilized powder): each bulb contains 10,000,000 IU recombinant human interferon alpha 2b; disodium hydrogen phosphate; sodium chloride; human serum albumin; dextran 40; mannitol 20%; Sodium dihydrogen phosphate dihydrate.
Heberon® Alfa R 3 M (solution for injection): each 0.3 mL bulb contains 3,000,000 IU recombinant human interferon alpha 2b; benzyl alcohol 3 mg; sodium chloride; polysorbate 80; monobasic sodium phosphate; dibasic sodium phosphate; water for injection.
Heberon® Alfa R 5 M (solution for injection): each 0.5 mL bulb contains 5,000,000 IU recombinant human interferon alfa 2b; benzyl alcohol 5 mg; sodium chloride; polysorbate 80; monobasic sodium phosphate; dibasic sodium phosphate; water for injection.
Heberon® Alfa R 10 M (solution for injection): each 1 mL bulb contains 10,000,000 IU recombinant human interferon alpha 2b; benzyl alcohol 10 mg; sodium chloride; polysorbate 80; monobasic sodium phosphate; dibasic sodium phosphate; water for injection.
Store at 2 to 8 ° C. Do not freeze or shake.
Heberon® Alfa R is beneficial in patients suffering from:
- Viral diseases: human papilloma virus infections, viral hepatitis, people infected with human immunodeficiency virus and dengue.
- Tissue neoplasms hematopoietic: hairy cell leukemia, chronic myeloid leukemia, multiple myeloma, low and medium malignancy non-Hodgkin lymphoma, cutaneous T lymphomas (mycosis fungoides, Sézary syndrome and others).
- Other hematological diseases: polycythemia vera, thrombocythemia and hypereosinophilia.
- Solid tumors: basal cell carcinoma of the skin, superficial carcinoma of the bladder, melanoma, carcinoid tumors, hemangioma of childhood, AIDS-associated Kaposi sarcoma, metastatic renal carcinoma.
- Fibrosis diseases: Peyronié’s disease, liver cirrhosis and keloids.
- < em> Diseases of the central nervous system: paranoid schizophrenia and multiple sclerosis.
Heberon® Alfa R is contraindicated in patients with hemoglobinopathies or hypersensitivity to interferon alfa or any of the salts present in the preparation.
See special warnings and precautions for use.
Heberon® Alfa R should be administered under the supervision of a specialist physician. Treatment may cause adverse reactions of moderate or severe intensity that require a dose reduction, temporary withdrawal of treatment, or permanent suspension.
- Cardiovascular system: Heberon® Alfa R should be administered with caution in patients with a history of severe heart disease. Although no direct cardiotoxic effects have been demonstrated, it is possible that some of the side effects (eg fever, chills, malaise), frequently associated with the administration of Heberon® Alfa R, may exacerbate a previous cardiac disturbance.
- Hematological system: Treatment with Heberon® Alfa R has been associated with a decrease in the total white blood cell count, the absolute neutrophil count and the platelet count, which generally begin in both first weeks of treatment. These decreases are reversible by reducing the dose or stopping therapy. Because one of the toxic effects associated with the use of Heberon® Alfa R may be leukopenia, extreme caution should be exercised when administering it to patients with myelosuppression.
- Renal system: Heberon® Alfa R should be administered with caution in patients with compromised renal function or a history of severe renal disorders; Since the kidney is the site where interferon alpha is metabolized, a reduction in the dose of the product should be considered, if necessary.
- Psychiatry and central nervous system (CNS): CNS adverse reactions have been reported in patients treated with interferon alfa at relatively high doses. These reactions include decreased mental alertness, aggressive behavior, confusion, and altered mental status. Most of these manifestations present with mild intensity and are reversible after dose reduction or discontinuation of interferon alpha therapy. Serious CNS effects associated with the use of interferon alpha, such as depression, suicidal ideation and suicide attempt, have also been observed in some patients during treatment and even for six months after its termination.
Heberon® Alfa R should be administered with caution in patients with a history of seizures or other CNS functional impairment. In patients with a psychiatric history, treatment with Heberon® Alfa R should only be started, after a specialized diagnostic evaluation has been carried out to rule out the active presence of any episode, sign or symptom of psychiatric disorders.
The course of treatment in this type of patient should be carefully monitored, and if the appearance of any of these manifestations is detected, the need for concomitant psychiatric treatment should be considered; If psychiatric symptoms persist or worsen or suicidal ideation is observed, it is recommended to immediately stop treatment with Heberon® Alfa R and monitor the patient with appropriate psychiatric treatment.
Endocrine system: With the use of alpha interferons, the appearance of thyroid function impairments or worsening of pre-existing thyroid diseases have been observed. Patients with uncontrolled hypo or hyperthyroidism should not start treatment with Heberon® Alfa R. Patients who develop some thyroid disorder during treatment and who cannot be controlled with concomitant medication or dose management of Heberon® Alfa R , they should temporarily interrupt treatment with Heberon® Alfa R until thyroid function returns to normal.
Autoimmune diseases: The use of different formulations of interferon alpha has been associated with an increase in allergic or autoimmune manifestations such as bronchoconstriction, lupus erythematosus, psoriasis, atopic dermatitis or thyroiditis. Although these conditions occur very rarely, Heberon® Alfa R should be administered with caution in patients with a history of autoimmune or allergic diseases.
Ophthalmology: Heberon® Alfa R has been reported very little associated with ocular changes in patients. However, alpha interferon has been associated with retinopathies, including retinal hemorrhages, cotton wool spots, papilledema, optic neuropathy, and retinal artery or vein obstruction, which can lead to loss of visual ability.
Pulmonary disorders: As with other alpha interferons, pulmonary symptoms, including dyspnea, pulmonary infiltrates, pneumonia, and pneumonitis have been reported during treatment with Heberon® Alfa R. In the presence of persistent or unexplained pulmonary infiltrates or lung function disorders , the treatment must be suspended.
Carcinogenesis: There is no experimental evidence or in the literature to suggest any carcinogenic potential. On the contrary, effects of Heberon® Alpha R have been found in the opposite direction, on differentiation in transformed cell lines and in tumors in animals.
Mutagenesis: None of the tests performed has shown or suggested that Heberon® Alpha R can be mutagenic. These include the Ames test with different strains and the test with human lymphocytes in cultures for chromosomal damage.
Pediatric use: Heberon® Alpha R has been used in children with type B and C viral hepatitis, recurrent respiratory and laryngeal papillomatosis, in hemangiomas, as well as in malignant and benign neoplasms. Side effects have been similar to those found in adults, mainly fever and general malaise. No disorders of growth or psychosomatic development have been reported, even after several months of continuous treatment.
Adverse reactions produced by interferon alfa are dose dependent and reversible. At higher doses the same adverse reactions occur as at lower doses, although with greater intensity. If this occurs, the dose should be reduced or treatment discontinued depending on the patient’s clinical situation. Although the general experience is that side effects diminish as the therapeutic regimen progresses, their continuation or restart must be carefully monitored.
The main side effects found with the use of Heberon® Alfa R are similar to those reported for other interferon alfa preparations. Its intensities are generally mild (does not require treatment) or moderate (responds to symptomatic treatment). Patients receiving doses ≥ 6 MIU may have adverse reactions with severe intensities that require additional measures, hospitalization, prolongation of hospitalization or suspension of treatment.
Frequency of the main adverse reactions of Heberon® Alfa R in an analysis of 2201 patients by the CIGB:
Headache (49%), fever (41%), asthenia (24%), myalgia (23%), anemia (20%), arthralgia (18%), chills (16%), leukopenia (15%) , anorexia (9%), nausea (8%); 4% frequency: malaise, weight loss, vomiting, depression, thrombocytopenia, insomnia, diarrhea, body aches, itching; 3% frequency: fatigue, dizziness, irritation, alopecia, tremors, abdominal pain, rash and hyperpolymenorrhea; frequency 2%: dry mouth; frequency 1%: drowsiness, cramps, and tachycardia; frequency 0.7%: neutropenia and mental confusion; frequency 0.6%: dyspnea, dry cough; frequency 0.5%: aphthous stomatitis, bone pain, and high blood pressure; frequency 0.4%: hypotension, dyspepsia, flu-like symptoms, odynophagia, eye irritation, decreased libido; frequency 0.3%: facial flushing, upper respiratory infection, blurred vision; frequency 0.2%: sweating, menstrual disorder, ocular pruritus, metrorrhagia, epitaxis, chest pain, tongue irritation, urinary sepsis, eyelid edema, impotence, hypoglycemia, lymphopenia; frequency 0.1%: back pain, anxiety, hypothyroidism, hypertransaminasemia, smell disturbances, granulocytosis, anisocytosis, macrocytosis, poikilocytiosis, macroplates, dysphonia, rash, dysgeusia, onychomycosis, dark urine, bronchial asthma, cystitis, dermatitis, meteorism, burning scalp, tongue pain, retroorbital burning; frequency 0.05%: spasticity, lipomas, subcutaneous nodules, hypersalivation, ringing in the ears, petechiae, stomatological disorder, nonspecific adenitis, neck pain, heartburn, eyelid ptosis, hyperuricemia, lymphangitis, monocytosis, hearing loss, mucositis, pneumonia, photosensitivity, puncture site hematoma, hyperthyroidism, bradycardia.
Recombinant interferon alpha preparations are immunogenic in a variable proportion of treated patients. The antibodies generated can neutralize the activity of interferon and, therefore, impede or reverse the response to treatment. The immunogenicity of Heberon® Alfa R has been monitored using blood samples taken from patients included in clinical trials carried out with this product. In 563 patients evaluated there were 14 (2.5%) with anti-interferon alpha antibodies with neutralizing capacity for antiviral activity. Heberon® Alfa R is less immunogenic compared to similar formulations on the international pharmaceutical market.
In adults, the standard dose is 3 to 6 MIU. In some particular situations of cancer patients, higher doses can be used, taking into account that the side effects will also be more intense. In children, the usual dose is 3 to 6 MIU per square meter (m 2 ) of body surface area. The frequency of administration varies between daily, cyclical (three times a week, every other day) and intermittent (1-2 times a week). The last two options are the most used in prolonged treatment regimens. The established route of administration is subcutaneous; however, intramuscular can also be used. Endovenous, intrathecal, intralesional, and intraperitoneal routes are equally possible.
Some of the useful treatment schemes are:
- Recurrent respiratory papillomatosis:
After surgical excision of the lesions, it is recommended to apply the scheme:
| Period | Children (IU / kg body weight) | Adults (MUI ) |
| 1 month | 100 000 3 v / s | 6 x 10 6 3 v / s |
| 1 month | 75,000 3 v / s | 3 x 10 6 3 v / s |
| 1 month | 50,000 3 v / s | 3 x 10 < sup> 6 3 v / s |
| 1 month | 50,000 2 v / s | 3 x 10 6 3 v / s |
| 8 months | 50,000 1 v / s | 3 x 10 6 2 v / s |
| 1 year | 50,000 month | 6 x 10 < sup> 6 mes |
If any relapse occurs, return to the immediate higher dose level or frequency.
Condyloma acuminata
6 MIU, three times a week, intramuscularly for six weeks.
3 MIU, three times a week, intramuscularly for six weeks, combined with cryosurgery of the lesions every 15 days.
Subacute viral hepatitis: 12 to 15 MIU for 3 to 5 days, intraperitoneally or intramuscularly, followed by 6 MIU intramuscularly every other day. This treatment is continued until the viral and disease markers are negative.
Acute liver failure in the infant: 3 to 6 MIU / m2 daily intraperitoneally for one week. The second week, the same dose intramuscularly. Depending on the patient’s evolution, the frequency of administration can be reduced to three times a week from the third week. Treatment is continued until viral and disease markers are negative.
Subacute (subfulminant) viral hepatitis in children older than one year: 3 to 6 MIU / m2 for one to two weeks: intraperitoneally the first week, and intramuscularly thereafter. Treatment can be continued with the same dose, three times a week, depending on the viral markers and the clinical situation.
Chronic hepatitis B:
In adults: 6 MIU daily intramuscularly for two weeks; then three times a week for four weeks, and twice a week for 16 weeks.
In children: 3 to 6 MIU / m2 intramuscularly three times a week for 16 weeks. If the child is older than 12 years, the dose should be 6 MIU.
Chronic hepatitis C: 3 MIU intramuscularly or subcutaneously, three times a week for 48 weeks. The effectiveness of the treatment increases when Heberon® Alfa R is combined with ribavirin in daily oral doses of 200 – 400 mg, three times a day for the same period.
People infected with the human immunodeficiency virus: 3 MIU, three times a week during the asymptomatic period. This treatment should be continued for a long time with hematological control, as well as monitoring of the possible development of anti-interferon alpha antibodies. In patients with Kaposi’s sarcoma a higher dose is recommended: 30 MIU / m2 / day, like other recombinant interferon alpha preparations. However, in cases where the diagnosis was made early, Heberon® Alfa R has induced total remissions with a dose of 6 MIU / day for six weeks.
Dengue: It should be used within 72 hours after the start of symptoms. In children, 50 to 100,000 IU / kg of body weight, daily, for three days. In adults, 3 to 6 MIU with the same frequency.
Chronic myeloid leukemia: Once hematological remission has been obtained, Heberon® Alfa R can be administered every other day, at a rate of 3 MIU / m2 until cytogenetic remission occurs, bone marrow transplantation can be performed or disease progression occurs .
Low- and medium-grade non-Hodgkin lymphomas: After obtaining remission by combination chemotherapy: 6 MIU, three times a week, for a year or more, until disease progression.
Basal cell carcinoma of the skin: In cases where surgical removal is not possible or not desired: 1.5 MIU, intralesionally, three times a week, for three weeks.
Superficial carcinoma of the bladder: Intravesical instillations of 20 MIU of Heberon® Alfa R in 50 mL, weekly for eight weeks and then monthly for two years.
Childhood hemangioma: In tumors larger than 2 cm in diameter, 3 MIU / m2, subcutaneously, daily for six months and then three times a week for approximately 12 months. This same dose can be applied intralesionally, twice a week, if the tumor is well defined.
Malignant melanoma: As an adjunct to surgery in patients without residual lesions but at risk of recurrence, 20 MIU should be used, five times a week, intravenously or intramuscularly, for four weeks. Subsequently, 10 MIU, three times a week, subcutaneously or intramuscularly, up to one year of treatment.
La Peyronié’s disease: A Heberon® Alfa R 10 MIU bulb is administered intralesionally, twice a week, for a maximum of 14 weeks, or less in those patients who have a complete response earlier.
Paranoid schizophrenia: A dose of 3 MIU of Heberon® Alfa R is administered twice a week and up to one year of treatment, adjunct to the usual treatment with neuroleptic drugs.
Multiple sclerosis: A dose of 10 MIU of Heberon® Alfa R is administered twice a week, intramuscularly for up to two years of treatment to patients with multiple sclerosis with the relapsing-remitting clinical form who have at least two relapses in the last two years. It is advisable to indicate prophylactically non-steroidal anti-inflammatory drugs or other antipyretics.
Heberon® Alfa R has synergistic action with some antitumor drugs in terms of the antiproliferative effect, which must be taken into account when applying a combination in the treatment of some neoplasms, since the myelosuppressive effect of both drugs would also be enhanced. Heberon® Alfa R also has synergistic action in both antiviral and antiproliferative effects, with gamma interferon. No drug interactions are reported.
Non-clinical studies in animal models with rats have not shown any teratogenic or fertility effects due to Heberon® Alpha R. There are no clinical studies to evaluate the effect of the product in pregnant patients; therefore, the safe use of Heberon® Alfa R during pregnancy cannot be established. Clinical experience with this product in pregnant women is limited to the treatment of 11 cases with thrombocythemia or chronic myeloid leukemia, where no alterations occurred, except for one case of low birth weight. Therefore, before using it, the doctor must do a risk-benefit analysis in each case.
The influence of Heberon® Alfa R on the ability to drive and use machines is small or moderate. If the patient is dizzy, confused, sleepy or fatigued, she should be advised that she should avoid driving vehicles or using machines. Overdose: Animal safety tests have shown tolerance to huge doses of Heberon® Alfa R, with no signs of toxicity.