Drug to control hyperinflammation, regulate the immune response and reduce the risk of mortality in patients with COVID-19 in serious or critical condition.
Lyophilisate for intravenous injection.
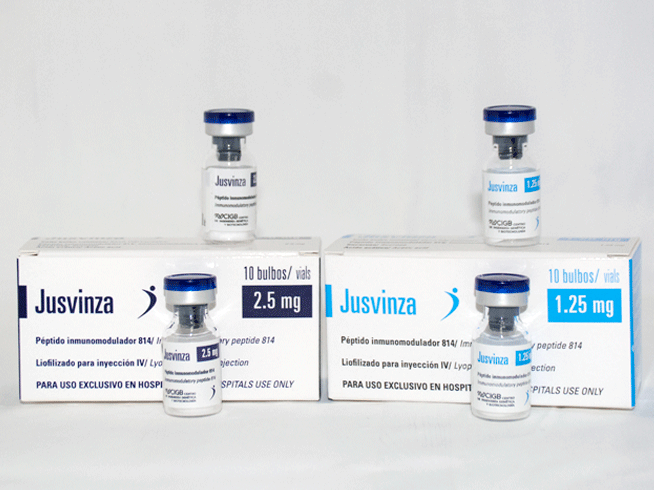
1.25 y 2.5 mg
Box of 10 colorless glass bulbs with 1.25 and 2.5 mg each.
Each vial contains:
- Immunomodulatory peptide 814 —— 1.25 or 2.5 mg
- Saccharose
- Glacial acetic acid
Store at 2 to 8 ° C (Refrigeration). Do not freeze.
- Jusvinza is indicated for the treatment of seriously and critically positive COVID-19 patients in whom a state of hyperinflammation is suspected or identified.
- It is used as part of the basic treatment for life support received by patients in serious or critical condition, in which antivirals, antibiotics, treatments for comorbidities, thromboprophylaxis or other specific interventions that are required are generally used.
- Jusvinza is contraindicated in individuals with hypersensitivity to any of the excipients of the formulation.
- It should not be used in patients receiving other biological medications, aimed at the treatment of cytokine storm, such as: anti-IL6R or IL-6 inhibitors (Tocilizumab), IL-1 inhibitor (Anakinra), JAK inhibitors and anti-CD6 (Itolizumab). Likewise, interferons should not be used in combination with Jusvinza in this medical condition
Use in pediatrics: Jusvinza has not been used in children, so its use in pediatrics is not recommended.
- Jusvinza is a medicine for hospital use and should be administered under the supervision of a specialist doctor.
- Immediate use (within 30 minutes of reconstitution), single dose.
Parenteral administration of this product is associated with a low probability of occurrence of adverse events. The safety profile has only reported pain and erythema at the injection site, skin rash and increased appetite, all with mild intensity and short duration.
- Jusvinza is administered intravenously.
- Patients WITH mechanical ventilation: 1 mg every 12 hours until extubation of the patient. Subsequently, it is continued with the same dose and route every 24 hours for three days. If at 72 hours there is no clinical, gasometric or radiological improvement, the dose may be increased to 2 mg, with a frequency of 12 hours (at the discretion of the medical practitioner). Never exceed 4 mg daily.
- Patients WITHOUT mechanical ventilation: 1 mg every 24 hours until the patient reverses his condition (that is, he does not require oxygen therapy and there are no signs and / or symptoms of acute respiratory failure). In case of unfavorable evolution and the need for mechanical ventilation, the Jusvinza therapeutic regimen is modified every 12 hours, detailed in the previous paragraph.
Steps to follow for the administration of 1 mg of Jusvinza:
Dilute, with a 1 mL syringe, the lyophilized powder contained in the bulb in the amount of water for injection defined according to the strength:
Strength (mg/vial) ——– Volume to add (mL)
1.25 ——————– 0.5
2.50 —————— 1.0
Shake gently until a clear, clear solution is obtained. Visually inspect the bulb; If any foreign particles and / or color change are observed, discard.
Using a 1 mL disposable syringe, load 0.4 mL and administer intravenously.
Steps to follow for the administration of 2 mg of Jusvinza:
- Dilute, with a 1 mL syringe, the lyophilized Jusvinza 2.5 vial in 1 mL of water for injections. Shake gently until a clear, clear solution is obtained. Visually inspect the bulb; If any foreign particles and / or color change are observed, discard.
- Using a 1 mL disposable syringe, load 0.8 mL and administer intravenously.
- Once the tablet has been dissolved in the injection water, it should be administered immediately and never exceeding 30 minutes. A Jusvinza 2.5 bulb can be used in two patients, provided that both coincide at the time of administration and that both patients have the 1 mg dose of the product indicated.
- Jusvinza is designed for immediate use after opening the bulb, therefore, at the end of each administration, the used bulb should be discarded with the remaining volume of solution. To prepare the next dose, a new bulb must be used.
Interactions with other medications have not been described.
There are no clinical studies conducted to evaluate the effect of the product in pregnant women, so its use is not recommended in this population group.
- It has not been described.
- In preclinical studies with the immunomodulatory peptide 814 for the evaluation of local tolerance in Lewis rats, it was shown that the repeated administration of the product, in a range of doses that exceeded up to 360 times the therapeutic dose, did not cause physiological or behavioral alterations in experimental animals. The clinical and macroscopic observations found no adverse or toxic signs attributable to the use of this peptide.