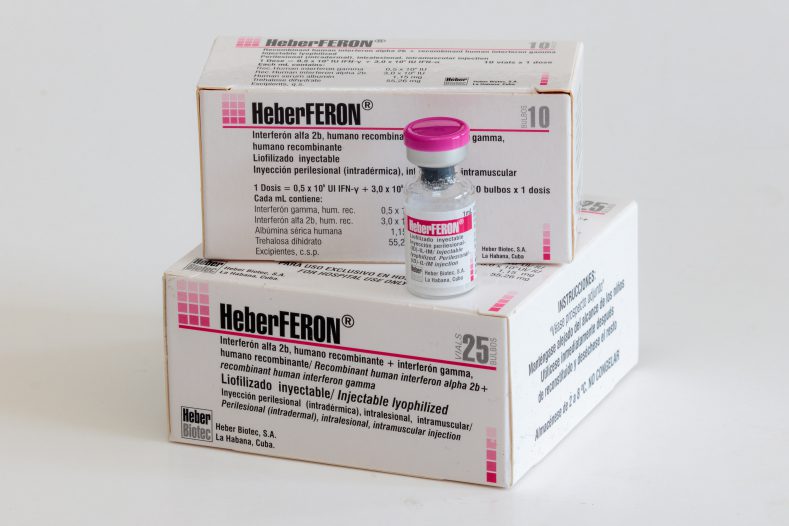
HeberFERON®
- Pharmaceutical combination in synergistic ratio of recombinant human interferon alpha 2b and recombinant human interferon gamma for the treatment of basal cell carcinoma.
- Treatment of lesions caused by basal cell carcinoma.
- HeberFERON® reduces tumor mass.
- Induce a rapid therapeutic response.
- Prolong response to treatment for at least 5 years.
- The frequency of the appearance of new lesions decreases by 8 times.
- Avoid complex surgeries in high-risk areas (periocular, ears, nose, scalp) and their sequelae.
- HeberFERON® improves the quality of life of patients. Leaves an excellent aesthetic at the injection site.
Lyophilisate for intralesional, intradermal and intramuscular injection.
5.0 x 10 5 UI IFN-γ + 3.0 x 10 6 UI IFN-α
- Box with 10 bulbs
- Box with 25 bulbs
Each 1 mL contains recombinant human alpha 2b interferon 3.0 x 10 6 UI; Recombinant human gamma interferon 5.0 x 10 5 IU; human serum albumin; trehalose dihydrate; succinic acid; water for injection.
Store between 2 and 8 ° C.
HeberFERON® is indicated for the perilesional (intradermal) or intralesional treatment of basal cell carcinoma confirmed by biopsy. It can be used as an alternative or adjunct treatment to other procedures (surgical or not), as well as in lesions of any size, clinical subtype and location, even high risk (such as in the H zone of the face) or locally advanced (such as lesions difficult to treat, due to local invasion and / or proximity to important structures such as the eyes and brain).
HeberFERON® is contraindicated in patients with hypersensitivity to any of the interferons (alpha or gamma) or to any of the ingredients present in the preparation. It is also contraindicated in patients with autoimmune diseases, since the administration of interferons can exacerbate them, and in patients with multiple sclerosis, since it has been reported that the administration of interferon gamma can cause their exacerbation.
HeberFERON® should be administered with caution to patients with a history of severe heart disease, severe kidney or liver disorders, seizures or other functional impairment of the central nervous system, and autoimmune or allergic diseases. As the occurrence of myelosuppression due to the use of interferons has been reported, the hematological status of patients receiving this drug should be monitored.
Although no direct cardiotoxic effect has been demonstrated, in patients with heart disease or a history of cardiac disorders, it is possible that some of the side effects (eg, fever, chills, headache) frequently associated with the administration of any of interferons, exacerbate a previous heart condition.The adverse reactions produced by HeberFERON® are reversible. If they occur, the dose should be reduced or treatment interrupted, as appropriate, and appropriate measures taken according to the patient’s situation. Although the general experience is that side effects decrease as HeberFERON® therapy continues, its continuation or restart in these cases should be carefully monitored.
This medicine can only be used until the expiration date indicated on the package. It should be used immediately after reconstitution. When adding the water to dissolve it, the formation of foam should be avoided, for which it is recommended that the liquid falls gently down the walls of the bottle. It should not be used if, once reconstituted, it presents a precipitate, turbidity or color. Do not reuse if content remains (discard).
The main side effects found in the use of HeberFERON® are similar to those presented by its components individually (alpha and gamma interferons), but of less intensity. These adverse reactions are reversible and dose dependent. Its intensity is generally mild (does not require treatment) or moderate (responds to symptomatic treatment). The main reactions in the cases studied were: fever (42.1%), chills (21.1%), arthralgia (5.3%), myalgia (26.3%), asthenia (15.8%), allergy (1.7%), pruritus (1.7%), weight loss (1.7%), thrombocytopenia (1.7%) and leukopenia (1.7%). Elevation of serum levels of liver or kidney damage markers has not been reported at doses below 20 MIU. Typical adverse reactions to interferons such as fever, headache, vomiting, drowsiness and other less common ones such as extrapyramidal reactions, aphasia and dysphasia have been observed intracranially. All reactions are reversible by reducing the dose and / or the frequency or discontinuation of the administration of the product. There have been episodes of transient cerebral ischemia in some cases with a history of this clinical situation, only in the lower eyelid area.
Calculate the amount of total product to use. Depending on the area of the lesion, distribute 1 mL (cc) of the total HeberFERON® dose of 10.5 MIU for every 1.5 cm 2 of the lesion surface.
Reconstitute 3 HeberFERON® bulbs with 1 mL (cc) of Water for Injections (USP). Add the diluent by letting it fall gently down the walls of the bottles, inverting them (without shaking and without foaming) until their contents are completely dissolved, for this use a 24G x 1½ ″ syringe with a needle.
Add the content of the first HeberFERON® bulb (extracted with the 24G x 1½ ″ needle) to the content of the second HeberFERON® bulb and in the same way to the third HeberFERON® bulb. If the calculated total volume to be injected was greater than 1 mL, transfer this volume of the mixture of the 3 HeberFERON® bulbs to a sterile bottle with a volume of 10 mL and bring it to the calculated total volume (required) with water for injection.
Apply the drug equidistant around the lesion, at a distance of 3 to 5 mm from the tumor edge (perilesional / intradermal). Insert the short or long needle 26 with the bevel upwards and at an angle of 15 ° (intradermal). Infiltrate the drug slowly until the wheal forms and achieve contact with the tumor periphery.
In patients with advanced basal cell carcinomas, HeberFERON® can be combined with chemotherapy cycles every 21 days (maximum 4 cycles). Chemotherapy doses must be calculated following the Calver procedure. They can be used as chemotherapy: cisplatin 50-100 mg / m 2 , carboplatin 100 mg / m 2 , adriamycin 50-70 mg / m 2 .
Administer HeberFERON® three times a week for 3 weeks perilesionally (intradermally).
Interactions with other drugs and other forms of interaction: HeberFERON® may have synergistic action with some antitumor drugs in terms of the antiproliferative effect, an aspect that must be taken into account when applying the combination in the treatment of some neoplasms, since it also the myelosuppressive effect of both drugs could be enhanced.
Pregnancy: HeberFERON® has not been used in pregnant women. There are also no studies with the use of recombinant human interferon gamma or interferon alpha in pregnant women. Therefore, its safe use during pregnancy has not been established and the doctor must do a risk-benefit analysis in each case before use.
Pediatric use: HeberFERON® has been used in children with hemangiomas without severe adverse reactions.
Effects on driving vehicles or machinery: The administration of recombinant human interferon alpha or interferon gamma can cause fever, decay and general malaise of varying intensity between 3 and 4 hours after the injection. These symptoms can affect the ability to drive vehicles and use machines.
Animal safety tests have shown tolerance to mega-doses of recombinant human interferon gamma with no signs of toxicity. Clinical tests have confirmed the good tolerability of this preparation even at doses as high as 20,000,000 IU / m 2 .
Adverse reactions to IFN alpha-2b are dose-dependent. At doses greater than 10 x 10 6 IU / m 2 of body surface area, the same adverse reactions occur, although with greater intensity. States of severe asthenia and depression have been described at very high doses. These effects are reversible.
In the case of HeberPAG® (combination of recombinant human interferon alfa 2b and recombinant human interferon gamma), safety tests in animals have shown adequate tolerance to high doses without any signs of systemic toxicity or adverse effects. HeberPAG® shows less intensity in its adverse reactions than IFN alfa-2b, at similar or higher doses.