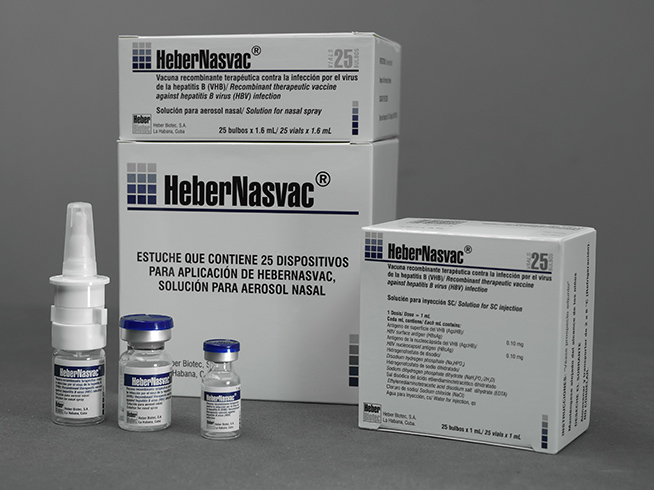
HeberNasvac®
Therapeutic recombinant vaccine against chronic hepatitis B virus infection. Vaccine for immunotherapy against chronic hepatitis B virus infection and prevention of its possible consequences. HeberNasvac® is administered through the mucosal (intranasal) and parenteral (subcutaneous) routes. HeberNasvac® induces a strong immune response.
Solution for nasal spray and solution for subcutaneous injection.
100 µg / mL of surface antigen of the hepatitis B virus (HBsAg) + 100 µg / mL of antigen of the nucleocapsid of the hepatitis B virus (AgcHB).
- Cases for 1; 5; 10 or 25 colorless glass vials with 1.6 mL each of nasal spray solution.
- Cases of 1; 5; 10 or 25 colorless glass vials with 1 mL each of solution for subcutaneous injection.
HeberNasvac® has the same composition in its nasal spray presentation as in the solution for subcutaneous injection.
Each 1 mL dose contains:
- 0.10 mg hepatitis B virus surface antigen (HBsAg)
- Hepatitis B virus nucleocapsid antigen (AgcHB) 0.10 mg
- Disodium hydrogen phosphate (Na2HPO4 )
- Sodium hydrogen phosphate dihydrate (NaH2PO4 x 2H2O)
- Disodium salt of ethylenediaminetetraacetic acid dihydrate (EDTA)
- Sodium chloride (NaCl)
- Water for injection q.s.
Store and transport at 2 to 8 ° C. Do not freeze.
- HeberNasvac® is indicated for active immunotherapy against chronic infection with the hepatitis B virus, and for the prevention of its possible consequences, such as liver cirrhosis, chronic liver failure and primary hepatocarcinoma.
- It should be applied to patients before the mentioned complications arise, since its usefulness and the risk-benefit ratio in those who already suffer from them have not yet been studied.
- HeberNasvac® is a therapeutic vaccine indicated only for adults and by prescription.
- Its use is not recommended in children under 18 years of age, as the clinical experience with this product, due to its novelty, is based exclusively on adults.
- It should not be administered to patients with febrile states, acute illnesses, or decompensated chronic ones, having to wait for the improvement of the clinical picture to start or continue treatment.
- Do not administer to people allergic to any component of the vaccine, or who have suffered a serious adverse reaction to a previous dose.
- HeberNasvac® should not be administered to patients with ALAT or ASAT enzyme levels above 500 IU/L.
- It should also not be administered to patients with hepatocellular carcinoma, liver cirrhosis or a history of liver transplantation.
- It is recommended to use with caution in cases of patients in whom chronic hepatitis B is concomitant with other liver conditions of another nature, such as alcoholism, autoimmune hepatitis, toxic hepatitis, Wilson’s disease, hemochromatosis and coinfection with the virus of the hepatitis C.
- HeberNasvac® produces a known transient increase in ALAT enzymes during or after treatment, which is associated with its mechanism of action. Regular monitoring of liver function should be considered during and after treatment.
- The safety and effectiveness of HeberNasvac® in pediatric patients have not yet been established.
- Not enough patients over 65 years of age have been evaluated to determine if they respond differently from younger individuals.
- In patients with compromised immune response, such as those receiving immunosuppressive therapies (e.g. corticosteroids, cytotoxic drugs, among others), in patients undergoing hemodialysis treatment and those suffering from immunodeficiencies due to HIV/AIDS or other causes, it could be affected the property of HeberNasvac® to achieve an effective immune response.
- Vaccination with HeberNasvac® of pregnant women is not recommended. The termination of pregnancy is not necessary in the event of unintended vaccination of the pregnant woman.
- As with any vaccine, a solution of epinephrine (1: 1000) should be available, ready for immediate use in the rare and unexpected event that an anaphylactic or other acute hypersensitivity reaction may occur.
- Although HeberNasvac® is a transparent and essentially homogeneous solution, some small particles of protein agglomerates can be observed in it, which are characterized as inherent particles of the product. The product should be discarded if a color change occurs.
- It should not be mixed in the bulb or syringe with any other vaccine.
- Do not administer intravenously.
- Clinical studies show that HeberNasvac® therapeutic vaccine is highly safe. Side effects occur in only a small number of people and are mild and short-lived.
- The most frequent events, according to the total doses administered, were: pain at the injection site (27.8%), sneezing (20.5%), runny nose (6.8%), slight local burning at the site of inoculation (6.8%), nasal pruritus (5.1%), slight fever less than 38 ° C (4.4%) and general malaise (4.4%). All these symptoms disappear spontaneously in the first hours without the need for treatment. In any case, it is recommended to consult a doctor when any undesirable manifestation appears.
- So far, in the studies carried out, no reactions of greater intensity or serious adverse events have been reported. In other hepatitis B vaccines, anaphylaxis and other types of immediate hypersensitivity reactions are reported in a very low proportion, which take place in the first hours after the administration of the vaccine.
- There is no confirmed scientific evidence that hepatitis B vaccines cause diseases of the central or peripheral nervous system, such as Guillain-Barré syndrome, optic neuritis, multiple sclerosis or other demyelinating diseases, chronic fatigue syndrome, rheumatoid arthritis or diseases autoimmune. It is recommended to evaluate the convenience of using an effective treatment for chronic hepatitis B and its sequelae through therapeutic vaccination against the risk, not scientifically confirmed, of inducing any of these diseases.
First cycle
HeberNasvac® for intranasal application by disperser: (0.10 mg of HBsAg + 0.10 mg of HBsAg) / 1.0 mL dose. A dose is applied every 2 weeks until 5 doses are completed.
Second cycle (one month after the end of the first cycle)
HeberNasvac® for intranasal application by disperser: (0.10 mg HBsAg + 0.10 mg HBG) / 1.0 mL dose and simultaneously HeberNasvac ® for subcutaneous injection: (0.10 mg HBsAg + 0.10 mg of AgcHB) / dose of 1.0 mL. One dose of each is applied, every 2 weeks until completing the 5 combined doses of both routes of administration.
- HeberNasvac ® is a therapeutic vaccine that has been shown to be safe and effective as monotherapy. Its simultaneous application with other specific therapies for the treatment of hepatitis B still needs to be studied.
- Co-administration with antivirals or immunomodulatory therapies such as interferon or pegylated interferon is not recommended.
Although the reactogenicity of HeberNasvac ® is very low, local symptoms such as pain at the injection site, sneezing and runny or itchy nose may appear, which, if they become severe in people Low thresholds may affect your ability to drive and use machines.
There are no reports to date of overdose with HeberNasvac ® . However, overdoses with the recombinant surface antigen, also used in vaccines to prevent hepatitis B, do not cause any special problems. Neither effects of overdose are expected with the recombinant antigen of the HBV nucleocapsid (AgcHB), nor with the other components of the formulation.