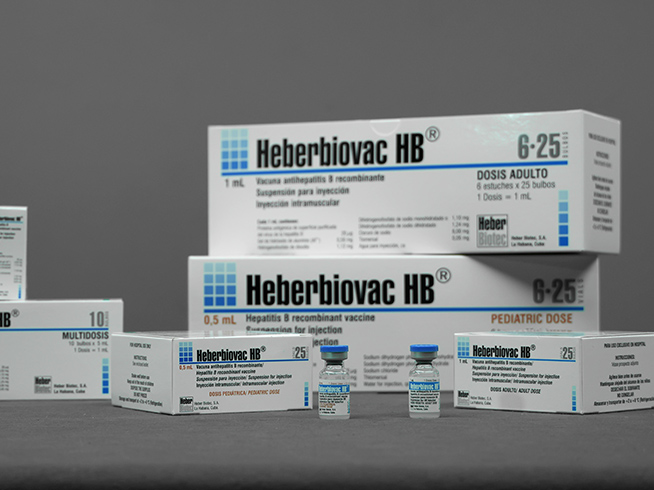
Heberbiovac HB®
Recombinant vaccine against hepatitis B virus and its consequences, Heberbiovac HB® is safe and immunogenic. It is a good ally in the control of delta hepatitis. Its production meets the requirements of the World Health Organization.
Suspension for intramuscular injection.
Each dose (1 mL) contains recombinant hepatitis B virus (AgsHB) surface DNA antigen: 20 µg.
- Cases of 1, 10 or 25 vials of 10 µg / 0.5 mL (pediatric dose)
- Cases of 1, 10 or 25 vials of 20 µg / 1 mL (doses for adults)
- Kits of 1 and 10 vials of 100 µg / 5 mL (multi-dose)
- Kits of 1 and 10 vials of 200 µg / 10 mL (multi-dose)
Each 0.5 mL dose contains:
- 10 μg recombinant HBV DNA surface antigen
- Aluminum hydroxide (Al 3+ )
- Sodium chloride
- Anhydrous dibasic sodium phosphate
- Monobasic sodium phosphate dihydrate or monobasic sodium phosphate monohydrate
- Thiomersal 0.05 mg
- Water for injection q.s.
Each 1 mL dose contains:
- 20 μg recombinant HBV DNA surface antigen
- Aluminum hydroxide (Al 3+ )
- Sodium chloride
- Anhydrous dibasic sodium phosphate
- Monobasic sodium phosphate dihydrate or monobasic sodium phosphate monohydrate
- Thiomersal 0.05 mg
- Water for injection q.s.
Temperature from 2 to 8 °C. Protected from light. Do not freeze.
Heberbiovac HB® is indicated for active immunization against hepatitis B virus (HBV) infection and the prevention of its potential consequences, such as acute and chronic hepatitis, liver cirrhosis, and primary hepatocarcinoma. It should be applied to all newborns and children who reach adolescence without having been vaccinated. It is particularly recommended in population groups at high risk of contracting the hepatitis B virus.
It should not be administered to individuals with febrile states due to severe infections, or to people allergic to any component of the vaccine, or who have suffered a severe adverse reaction to a previous dose. The vaccine will not harm individuals who have been or are infected with the hepatitis B virus.
As with any vaccine, a solution of epinephrine (1: 1000) should be available, ready for immediate use in the rare and unexpected case in which an anaphylactic reaction or another acute hypersensitivity reaction.
Due to the long incubation period of hepatitis B (up to 6 months or more), the disease may not be prevented if it is being incubated at the time of vaccination. This vaccine is not capable of preventing hepatitis caused by agents other than hepatitis B virus (such as hepatitis A, C and E viruses), but it is considered to be effective in preventing hepatitis caused by the delta agent. Do not administer intravenously.
Several studies have shown that the Heberbiovac HB ® vaccine is highly safe. The adverse effects that are temporarily associated with its administration are low frequency, mild and of short duration. In a post-marketing study that evaluated 40,533 doses administered to children under one year of age, the most frequent effects, according to the total doses administered, were pain at the site of inoculation (0.15%), mild fever less than 38 °C (0.14%) and redness at the injection site (0.10%).
In controlled clinical trials in children, adolescents and adults, the above symptoms have also been reported as the most frequent, and to a lesser extent limited induration, among local symptoms; and fever, headache, and weakness, among the systemic symptoms.
No relationship between more serious reactions and Heberbiovac HB ® vaccine has been established. In other hepatitis B vaccines, anaphylaxis and other types of immediate hypersensitivity reactions are reported in a very low proportion, which take place in the first hours after the administration of the vaccine.
There is no confirmed scientific evidence that hepatitis B vaccines cause diseases of the central or peripheral nervous system such as Guillain-Barré syndrome, optic neuritis, multiple sclerosis or other demyelinating diseases, chronic fatigue syndrome, rheumatoid arthritis or autoimmune diseases . It is recommended to evaluate the convenience of preventing hepatitis B and its sequelae through vaccination against the risk, not scientifically confirmed, of inducing any of these diseases.
- For newborns and children up to the age of 10 years: doses of 10 µg are recommended.
- For children over 10 years of age and adults: doses of 20 µg are recommended.
Two treatment schemes are recommended
- Two doses separated by 1-month intervals, followed by a third dose 6 months after the first dose (0-1-6).
- Three doses separated by 1-month intervals (0-1-2). This scheme is used in people at high risk of contracting the disease.
In immunosuppressed patients (for example, patients on hemodialysis), higher doses are required.
Doses of 40 μg (2 mL) are recommended using the scheme of three doses separated by intervals of 1 month and a booster dose at 6 months (0-1-2-6).
Booster dose
Follow-up of cohorts of vaccinated patients showing seroprotection for more than 10 years after vaccination indicates that the booster dose is not required, at least during that time, in people who received either of the two administration schedules Recommended.
Mode of administration
Heberbiovac HB ® vaccine should be administered by deep intramuscular injection in the anterolateral aspect of the thigh in newborns and children under 1 year of age. , or in the deltoid region of children older than 1 year or adults. Injection into the buttock is not recommended. Nor is it recommended intradermal injection, or intravenous, or other forms of administration. Shake the container before use.
Heberbiovac HB ® can be administered safely and effectively at the same time as BCG, DPT, Haemophilus influenzae type b, meningococcal BC, measles, oral or injectable polio (OPV or IPV), yellow fever and vitamin A supplements. In cases of simultaneous administration with other vaccines, it should be applied at different injection sites and with separate syringes. It should not be mixed in the vial or syringe with any other vaccine, unless it has been produced as a combined product (example: DTP-HB). It is interchangeable with other hepatitis B vaccines of a plasma or recombinant nature.
Vaccination of pregnant women is not recommended, but in high-risk situations or other special cases, the doctor may consider administration. The interruption of pregnancy is not necessary in the event of unintentional vaccination of the pregnant woman.
Although the reactogenicity of this vaccine is very low, fever and local symptoms may appear, which if they became intense in people with a low threshold, could affect your ability to drive and use machines.
Not reported.