Our vaccine against COVID-19 Abdala showed high levels of seroconversion (appearance of antibodies against covid-19) in children and adolescents between 3 and 18 years of age, who participated as volunteers in the Ismaelillo clinical study, in the province of Camagüey.
The group of infants up to 11 years old registered an increase in antibody titers of 99.15%, while those between 12 and 18 years old reported an increase of 98.28%.
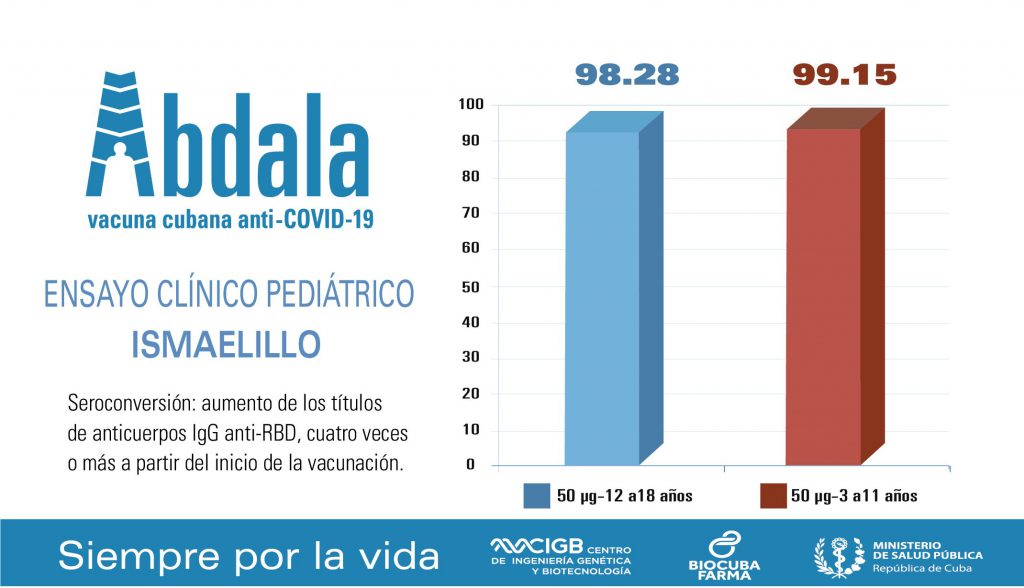
The increase in antibody titers was four times or more from the first dose of the immunogen, whose complete schedule is 3 doses, with an interval of 14 days between each administration.
The clinical trial concluded on October 14th. In this study 500 children participated and two immunogen strengths were used: 25 and 50 micrograms, in the same administration schedule 0-14-28 days.
The main researcher of the studies with this vaccine, Dr. Sonia Resik, Head of the Virology Department of the Pedro Kouri Institute of Tropical Medicine, highlighted at that time that more than 80% of the adverse reactions reported in children were mild and the rest of the indicators were comparable with the results in adults, which confirmed their effectiveness.
On October 27, the Cuban regulatory authority for medicines, equipment and medical devices, Cecmed approved the Authorization for the Emergency Use of the Abdala vaccine from 2 years of age, while its quality, safety was demonstrated and effectiveness.
Since July 19, Abdala obtained authorization for its use in emergency in adults in Cuba, after demonstrating an efficacy of 92.28% in the prevention of symptomatic covid-19, and became the first immunogen in Cuba and Latin America.
It also proved 90% effective in seriously ill patients affected by the SARS-CoV-2 coronavirus, which causes this condition.
This vaccine is also licensed for emergency use in Vietnam, Venezuela and Nicaragua.


comments on