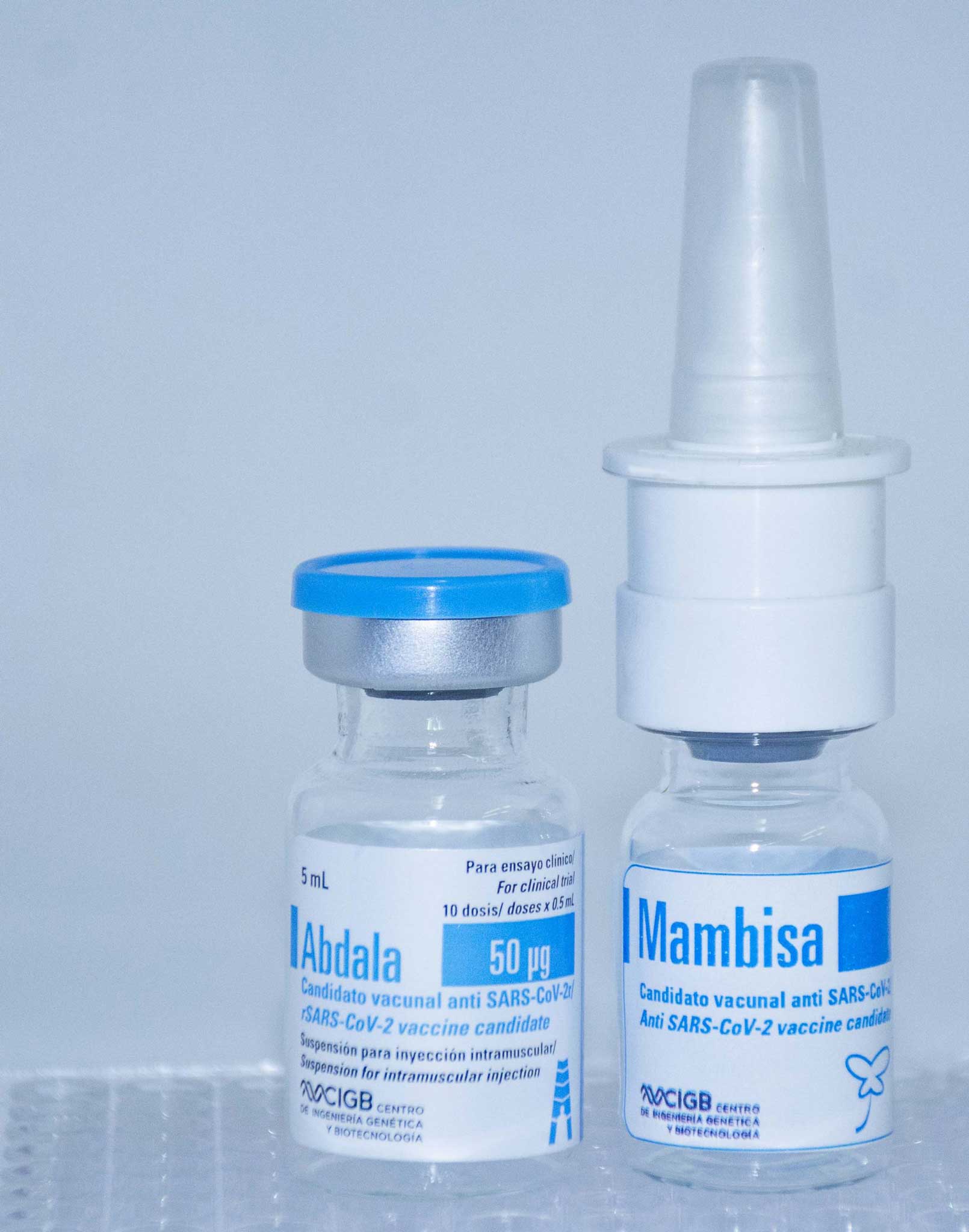
The second clinical trial in convalescent covid-19 developed in Cuba with the Abdala vaccine and the vaccine candidate Mambisa began this July 21, at the Hermanos Ameijeiras Clinical Surgical Hospital in Havana.
According to information released on Cuban television, the sample for this study is made up of 120 subjects who suffered from the disease and have been discharged for at least two months, belonging to the capital city of Centro Habana, aged between 19 and 80 years.
This is a phase I / II clinical trial, in which the patients will be divided into four randomization groups: three will be administered Mambisa and one Abdala.
Authorities Cuban regulators detailed that they seek to evaluate the safety and immunogenicity of both products, designed by the Center for Genetic Engineering and Biotechnology (CIGB).
According to the Cuban public registry of clinical studies, one of the objectives of this The stage is to verify the immunogenicity and functionality of the administration by nasal spray and by drops of the Mambisa product.
With this objective, the participants in each group to which Mambisa is administered will receive the product through e nasal spray, drip or through a national device adapted to a syringe.
In this way it is intended to evaluate, according to the benefit-risk-cost balance, which of the three is more effective and is the to be used in phase II of the study to continue with the evaluation of patients.
They will also evaluate the immunogenicity and functionality of the intramuscular administration of the vaccine Abdala, the first in Latin America and the Caribbean, which demonstrated a 92.28% efficacy in its three-dose schedule.
According to the public registry, it is expected with this clinical trial that 55% or more of the subjects treated with both vaccination proposals, increase in four times the initial determination of the specific antibody titers against the SARS-CoV-2 virus, the pathogen that causes covid-19.
They also foresee that the inhibition titers of binding to the ACE2 receptor, without the risk of more than 5% of individuals with serious adverse events with causality consistent with vaccination.
Havana, Jul 21 (Prensa Latina)

comments on